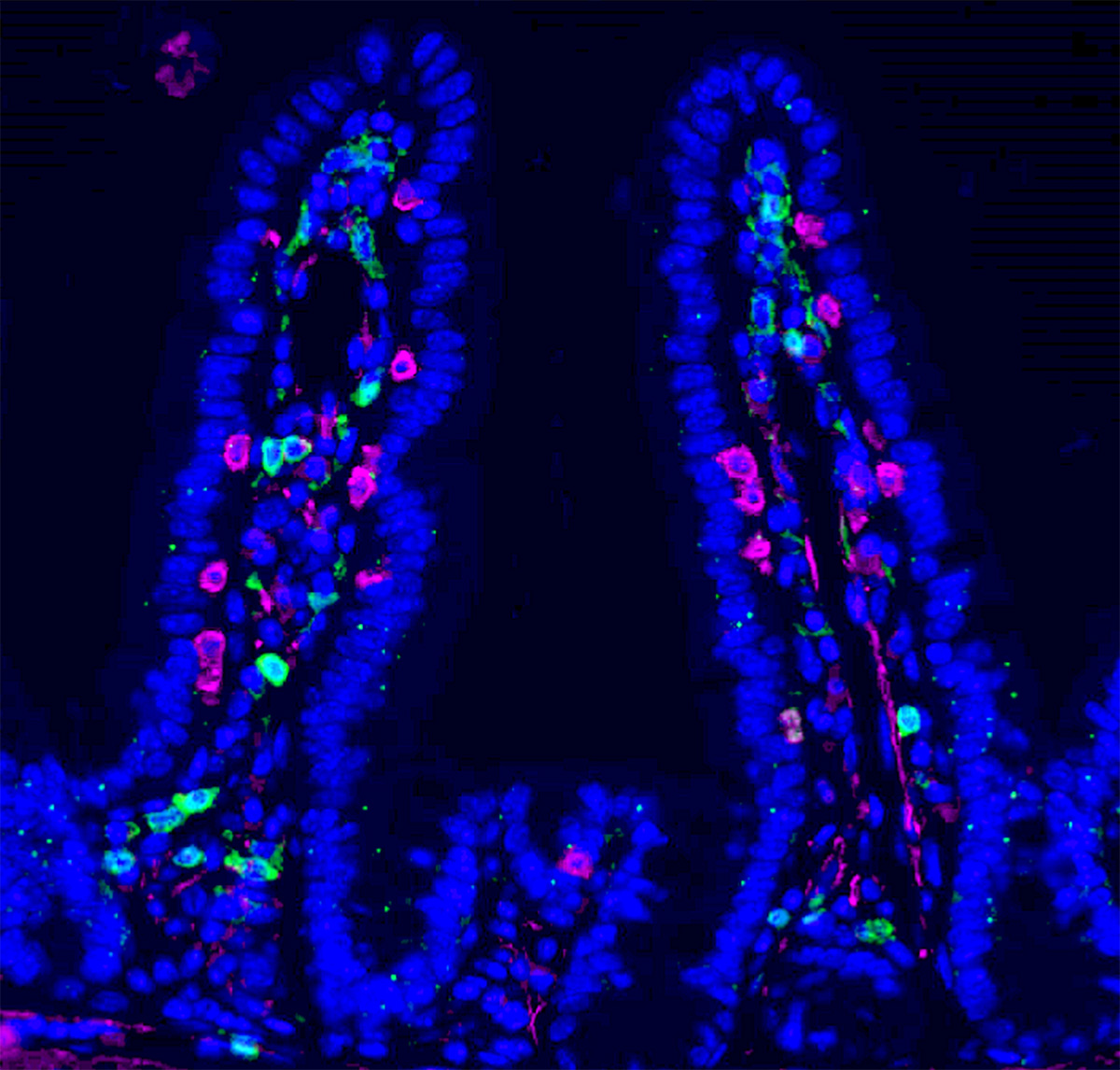
Researchers led by Ludwig Lausanne’s Ludivine Bersier and Tatiana Petrova reported in a December publication in Nature Communications a gut-to-bone marrow axis of signaling that helps prevent liver metastases of colorectal cancer (CRC) and is induced by the indirect effects of chemotherapy on gut microbes. They show that intestinal inflammation, or mucositis, caused by chemotherapy alters nutrient availability in a manner that promotes the growth of bacteria that metabolize tryptophan into indole-3-propionic acid (IPA). This molecule reprograms immune cell generation in the bone marrow to favor the development of macrophages over immunosuppressive monocytes, effectively giving a surprising, positive spin to a common side-effect of chemotherapy. The immune shift enhances the anti-tumor activity of CD4+ T cells and spatially reorganizes their interactions with CD8+ T cells at metastatic sites. Moreover, the observed reprogramming of the immune system outlasts the duration of therapy, disfavoring the post-treatment spread of the cancer to the liver. Ludivine, Tatiana and their clinical colleagues also showed that IPA levels in CRC patients are a fifth of those seen in healthy people and higher circulating IPA levels after chemotherapy in a subset of patients correlated with fewer monocytes and better outcomes. This suggests IPA might have therapeutic potential as an agent that prevents relapse in patients receiving chemotherapy.
Chemotherapy-driven intestinal dysbiosis and indole-3-propionic acid rewire myelopoiesis to promote a metastasis-refractory state
Nature Communications, 2025 December 15