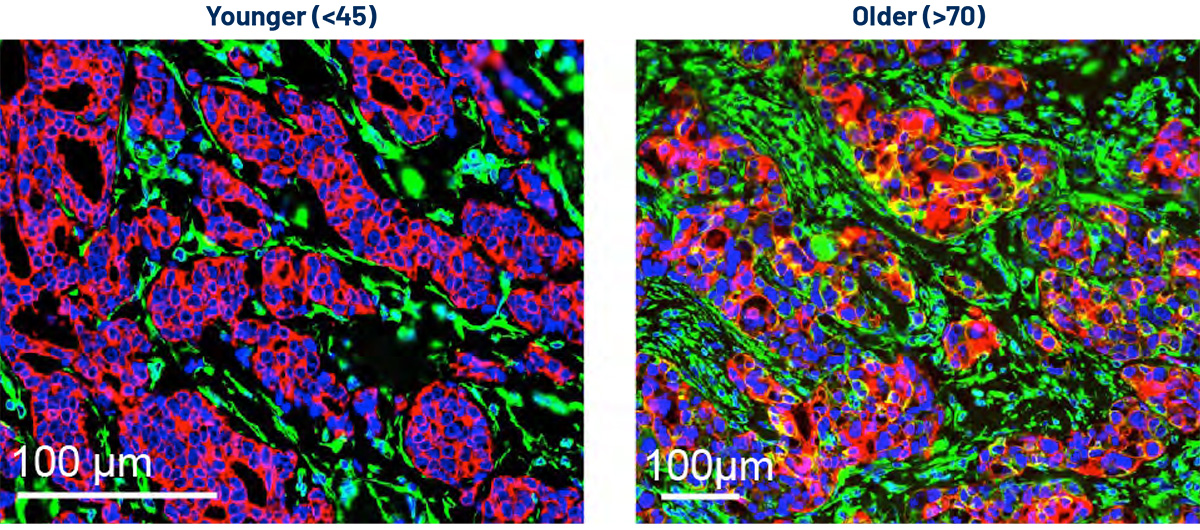
Compared to women between the ages of 55 and 64 who are diagnosed with breast cancer, women both younger and older have worse outcomes, regardless of their particular subtype of the cancer. Why this should be is unclear. Both demographics have long been underrepresented in clinical trials, and not much is known about how age affects the cells of the tumor microenvironment and shapes pathology. Yet the subtypes of disease seen most often in each subgroup do vary with age. The incidence of triple-negative breast cancer (TNBC), for example, is relatively higher in younger women, while hormone receptor-positive disease is markedly more common in older women. Through analysis of existing bulk and single-cell transcriptomic data, researchers co-led by Ludwig Harvard’s Peter van Galen and Harvard colleague Sandra McAllister developed a framework for understanding cell-specific, age-associated changes in gene expression, protein levels and cellular interactions within the tumor microenvironment in TNBC and estrogen receptor positive (ER+) breast cancers. They reported in a Nature Aging paper in November an Age-Specific Program ENrichment (ASPEN) analysis pipeline that exposes age-related changes in the malignant and noncancerous cells of the tumor microenvironment for both types of breast cancer, and the signaling pathways potentially underlying those changes. The study identified possible targets for age-adapted therapeutic interventions for breast cancer.
Cell populations in human breast cancers are molecularly and biologically distinct with age
Nature Aging, 2025 November 4