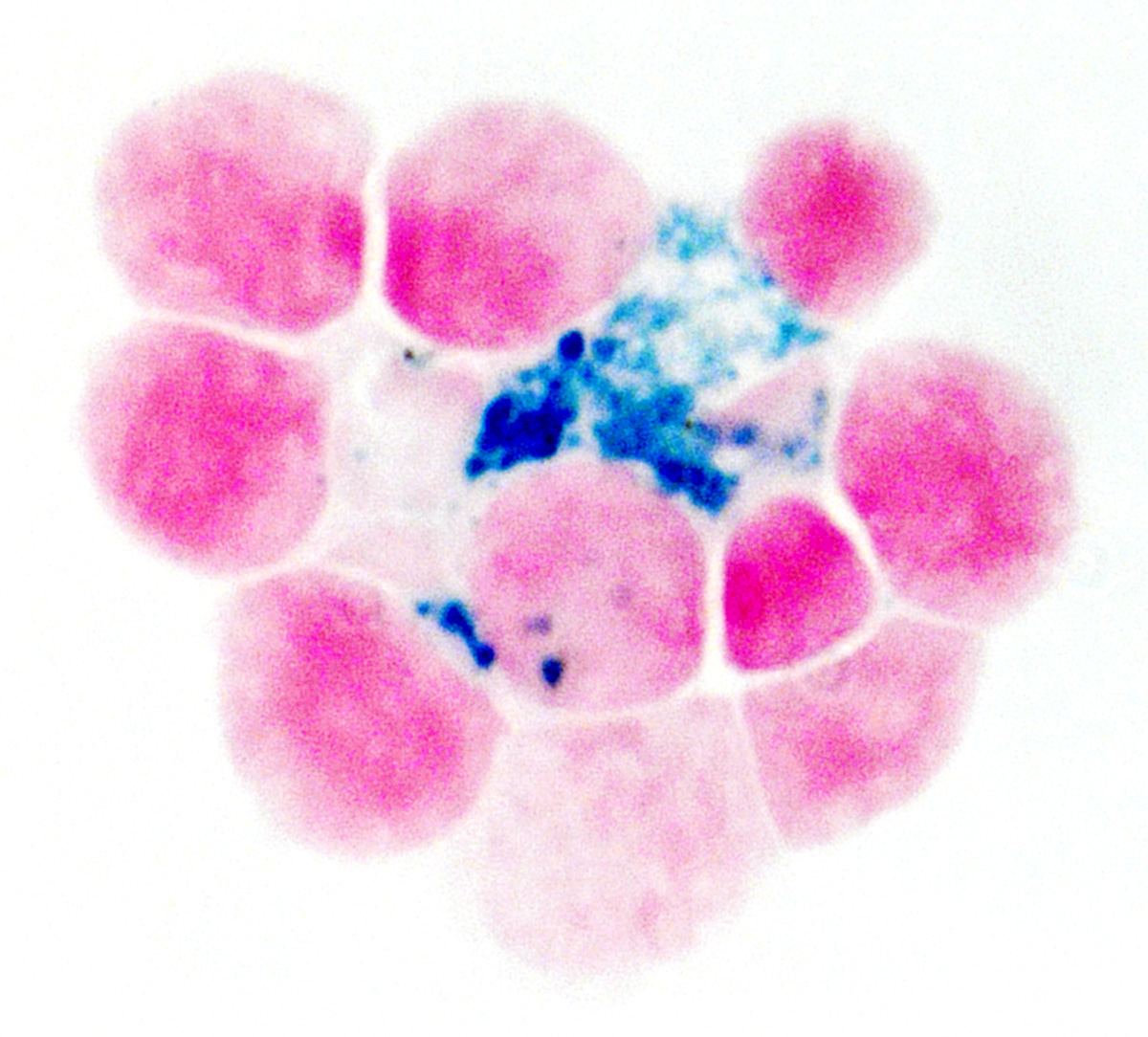
Breast cancers frequently spread to the bone, establishing tumors that are largely impervious to treatment and are associated with poor patient prognoses. Anemia is among the most common complications of such metastases. Researchers led by Ludwig Princeton’s Yibin Kang and Yujiao Han described in a Cell paper in September a pair of canny strategies breast cancer cells employ to access metabolic support in the oxygen-poor microenvironment of bone marrow that contribute to that anemia. They discovered that specialized iron-recycling immune cells—erythroblast island (EBI) macrophages—cluster around cancer cells in the metastatic niche in bone. Although bone marrow has healthy reserves of iron, an essential mineral, it is not readily available to invading cancer cells. EBI-macrophages ordinarily provide iron to erythroblasts, the precursors of red blood cells. Cancer cells, the researchers found, hijack EBI macrophages to acquire iron, disrupting the generation of red blood cells. Having hoarded the available iron, the cancer cells then begin mimicking erythroblasts, expressing a component of hemoglobin—β-globin—to better survive the hypoxic environment in the bone marrow. Together, these adaptations likely synergize to induce anemia. The researchers show that similar iron-handling macrophages are found in human bone metastases from lung and kidney tumors as well. This suggests that the hijacking of iron-recycling macrophages might be a common phenomenon in cancers that metastasize to the bone.
Tumors hijack macrophages for iron supply to promote bone metastasis and anemia
Cell, 2025 September 3