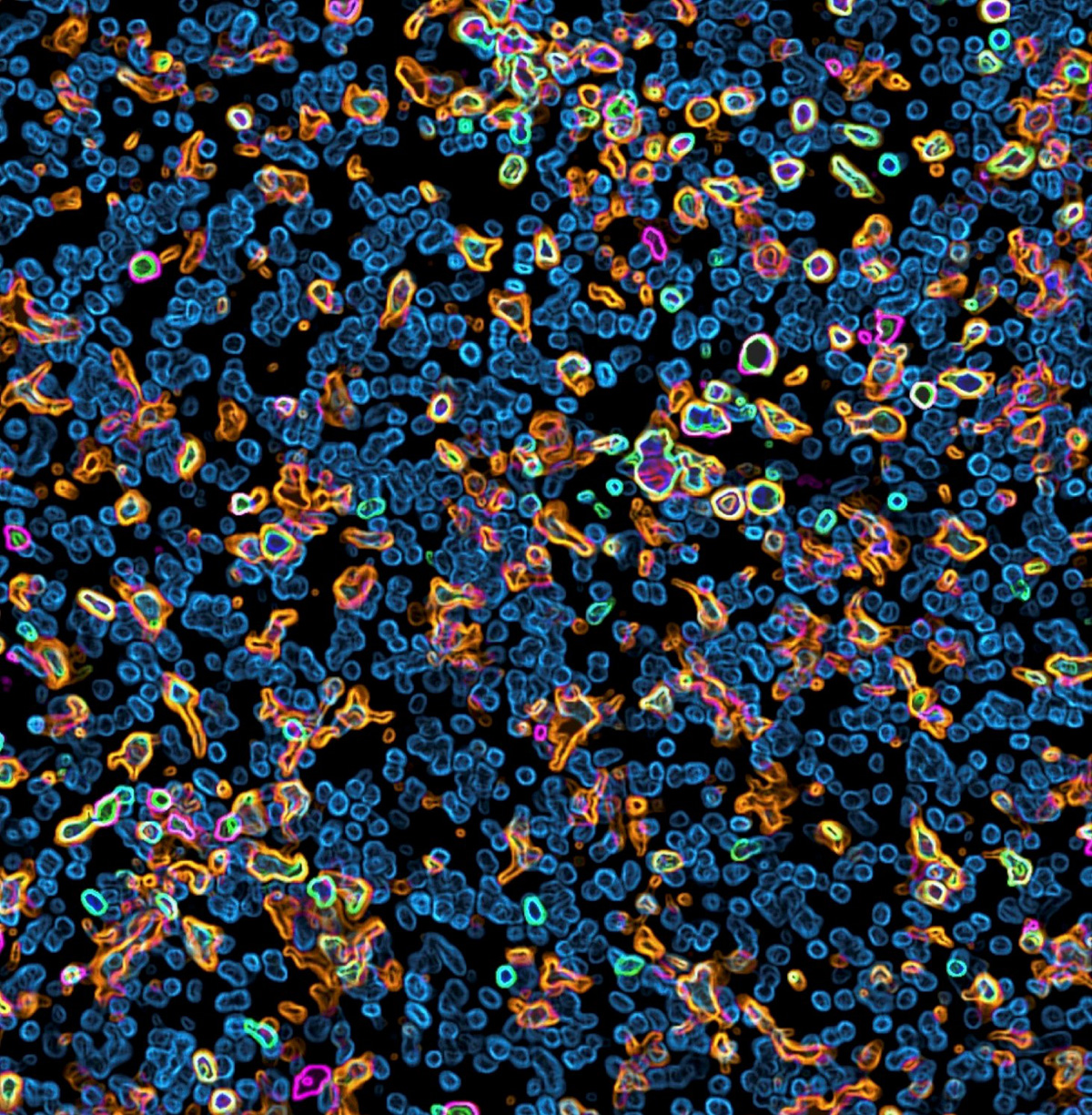
Glioblastoma multiforme (GBM) is notoriously resistant to treatment. This stems, in part, from the dizzying genetic variability of glioblastoma cells and the profoundly immunosuppressive microenvironment the tumor cultivates. Researchers led by Ludwig Lausanne’s Johanna Joyce and recent alumnus Ángel Álvarez-Prado identified in a September publication in Cell Reports a promising new drug target in GBM cells that could potentially address both these sources of resistance. Recent studies have found that certain types of cancer cells that continuously express interferon-stimulated genes (ISGs) are highly vulnerable to ADAR1 loss, and that deleting its gene in melanoma tumors can improve responses to immunotherapy in mouse models. Ángel, Johanna and colleagues showed that loss or chemical inhibition of ADAR1—which is a regulator of the innate antiviral response induced by type I interferons in mammalian cells—stalls the proliferation of genetically diverse ISG-expressing GBM cells in cultured human tumor samples and in mouse models. Further, disabling ADAR1 amplified ISG-stimulated inflammatory responses, which had the effect of reprogramming the tumor microenvironment of multiple, genetically distinct GBM tumors. This boosted the numbers of anti-tumor immune cells in the TME—such as CD8+ T cells, pro-inflammatory macrophages and natural killer cells—and depleted immune cells that suppress anti-tumor immune responses. ADAR1, the researchers note, is especially enticing as a target because its disruption delivers a one-two punch against the GBM tumor.
Cancer cell and microenvironmental rewiring by ADAR1 loss impairs glioblastoma tumor growth and extends survival
Cell Reports, 2025 September 1