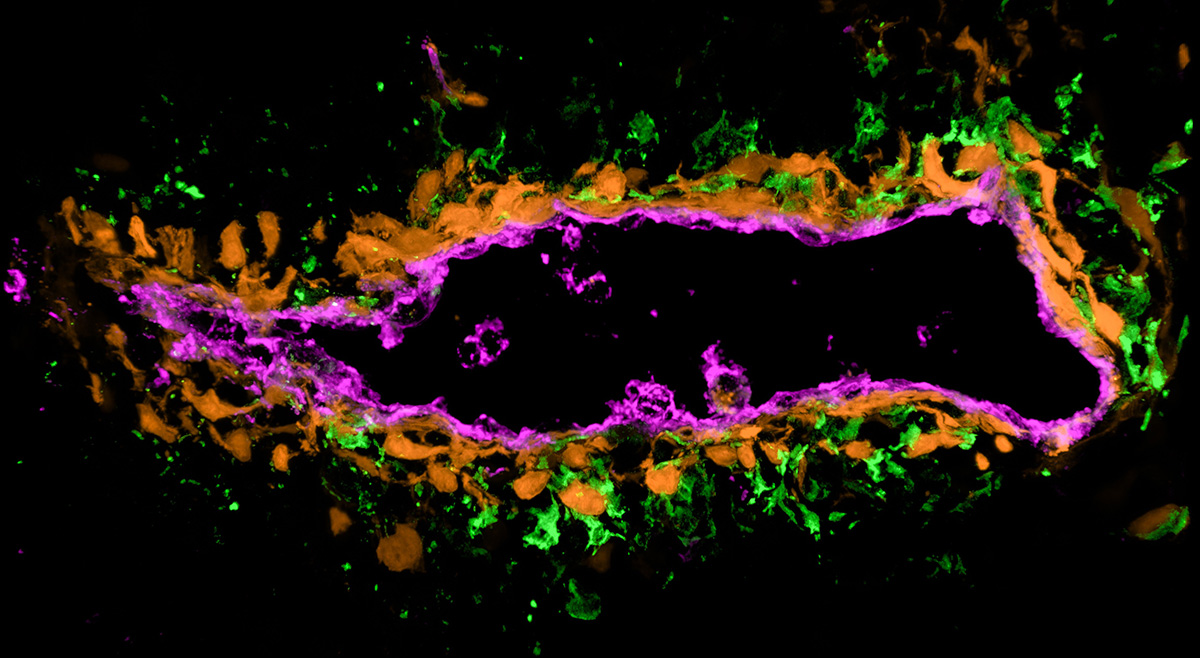
Researchers led by Ludwig Lausanne’s Mikaël Pittet described in a December paper in Immunity the localization of CCR7+ conventional dendritic cells (cDCs) within tumors and showed how their positioning influences anti-tumor immune responses. The chemokine receptor CCR7 is expressed by activated type 1 and 2 cDCs, professional antigen-presenting immune cells that activate and direct anti-tumor T cell responses. Their relative abundance in tumors is associated with better survival outcomes and responses to immunotherapy in cancer patients. Single-cell, spatial and intravital analyses of tumors from patients and in mouse models revealed that fibroblasts surrounding venous blood vessels produce the chemoattractant CCL19 to guide CCR7+ DCs into perivascular niches, where they cluster and activate T cells. CCR7+ DCs in these clusters are often contacted by regulatory T cells (Tregs), which suppress their expression of CD40 and inhibit their ability to activate CD4+ and CD8+ T cells. Mikaël and colleagues showed that depleting Tregs reverses this effect and improves T cell-mediated tumor control. Notably, anti-PD-1 immunotherapy promotes not only CCR7+ DC clustering but also interaction with Tregs via a CCL22-mediated mechanism, mitigating the efficacy of checkpoint blockade therapy. The authors suggested that the pharmacologic targeting of these cellular interactions in perivascular immune hubs may offer a path to improving patient responses to cancer immunotherapy.
Positioning and reversible suppression of CCR7+ dendritic cells in perivascular tumor niches shape cancer immunity
Immunity, 2025 December 19