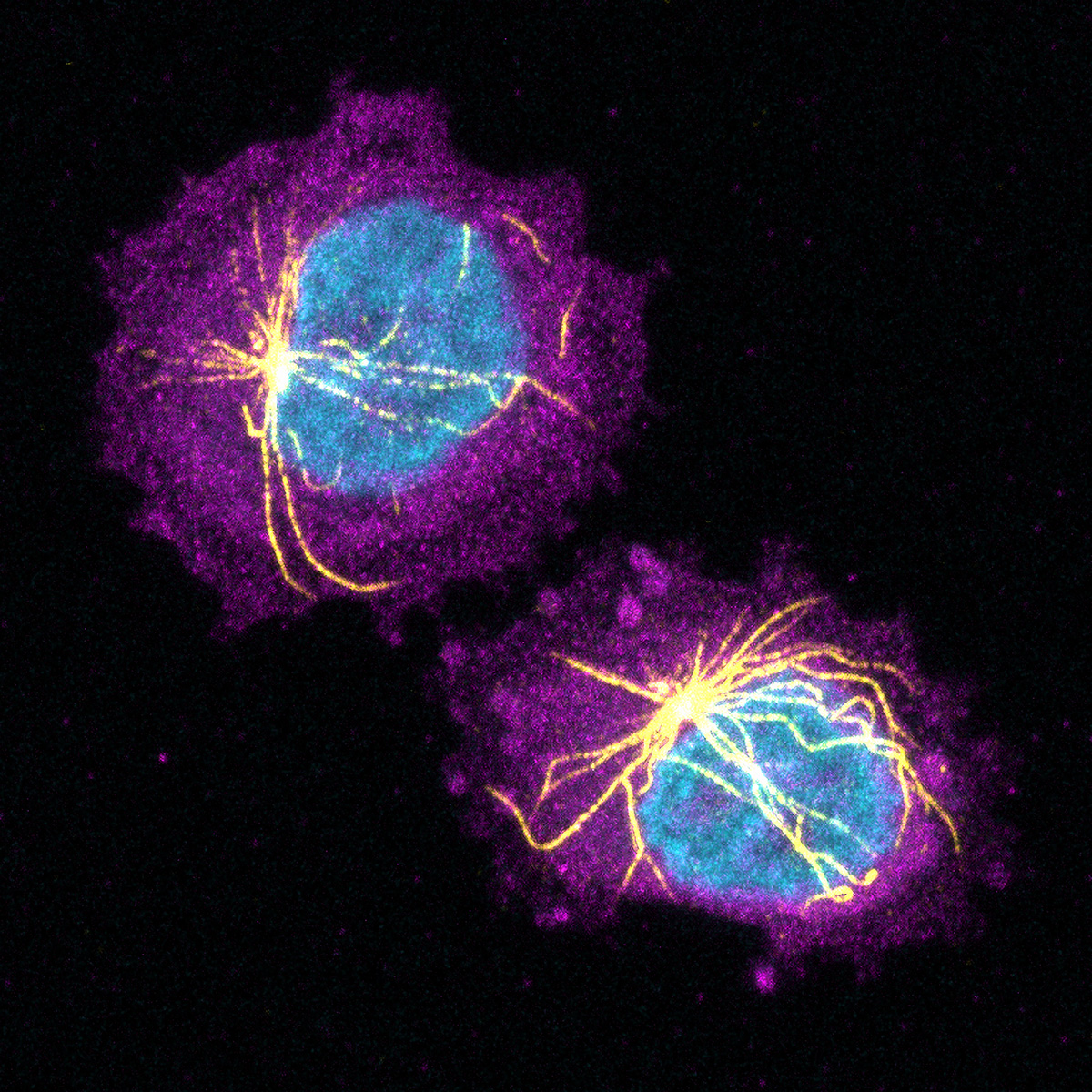
Researchers led by Ludwig Oxford’s Richard White and Miranda Hunter of the Memorial Sloan Kettering Cancer Center reported in an August paper in Nature how the tight physical confinement of tumor cells by surrounding tissues has epigenetic consequences that can influence melanoma progression. Rather than continuing to divide rapidly, cancer cells in such circumstances activate a program of ‘neuronal invasion’ and spread into neighboring tissues. Using a zebrafish model of melanoma, the researchers showed that HMGB2, a DNA-bending protein, responds to the mechanical stress of confinement by binding to chromatin, altering its structure and exposing regions of the genome encoding genes linked to invasiveness. Cells with high levels of HMGB2 thus become less proliferative but more invasive and resistant to treatment. They also remodel their cytoskeleton, forming a cage-like structure around the nucleus. This protective shield involves the LINC complex, a molecular bridge that connects the cell’s skeleton to the nuclear envelope, helping to protect the nucleus from rupture and DNA from damage caused by confinement-induced stress. The findings highlight an underappreciated dimension of microenvironmental influence on cancer cell behavior, showing how physical cues can prompt them to adapt their skeletal and genomic architecture to shift between states of growth and invasion. Most notably, however, the study also demonstrates how physical stress is a potent—and barely explored—driver of epigenetic change.
Mechanical confinement governs phenotypic plasticity in melanoma
Nature, 2025 August 27